
Visualizing Formation of Intermetallic PdZn in a Palladium/Zinc Oxide Catalyst: Interfacial Fertilization by PdHx - Niu - 2019 - Angewandte Chemie International Edition - Wiley Online Library

Mild Palladium-Catalyzed Cyanation of (Hetero)aryl Halides and Triflates in Aqueous Media | Organic Letters

Picolinaldehyde‐Zinc(II)‐Palladium(0) Catalytic System for the Asymmetric α‐Allylation of N‐Unprotected Amino Esters - Li - 2023 - Chemistry – A European Journal - Wiley Online Library
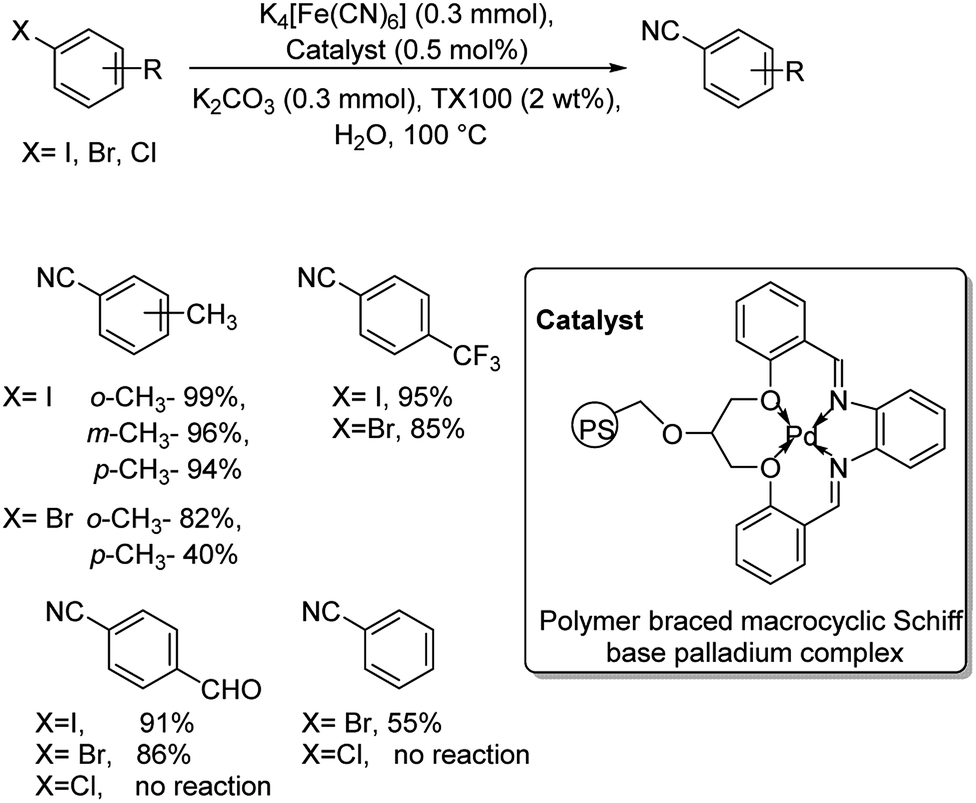
Recent advances and prospects in the palladium-catalyzed cyanation of aryl halides - RSC Advances (RSC Publishing) DOI:10.1039/D0RA05960A

Mild Palladium-Catalyzed Cyanation of (Hetero)aryl Halides and Triflates in Aqueous Media | Organic Letters
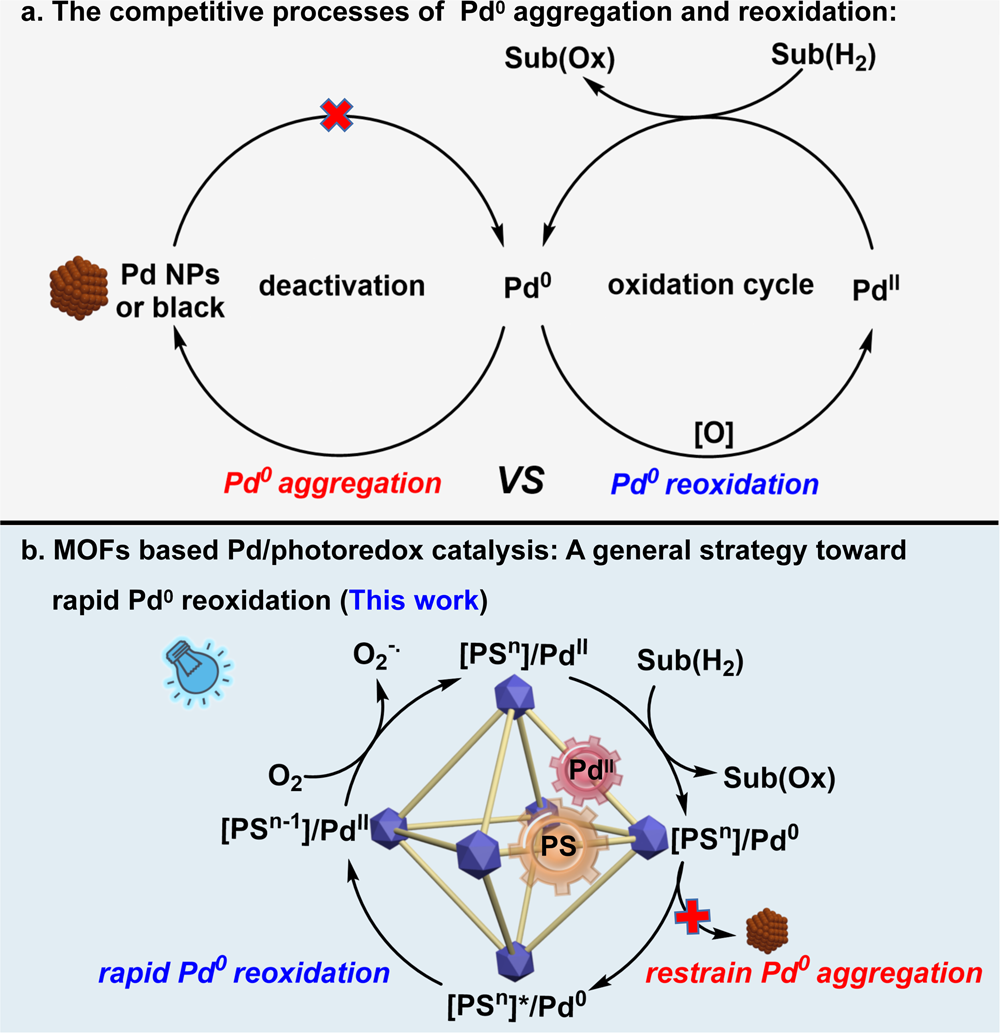
Visible light-driven efficient palladium catalyst turnover in oxidative transformations within confined frameworks | Nature Communications
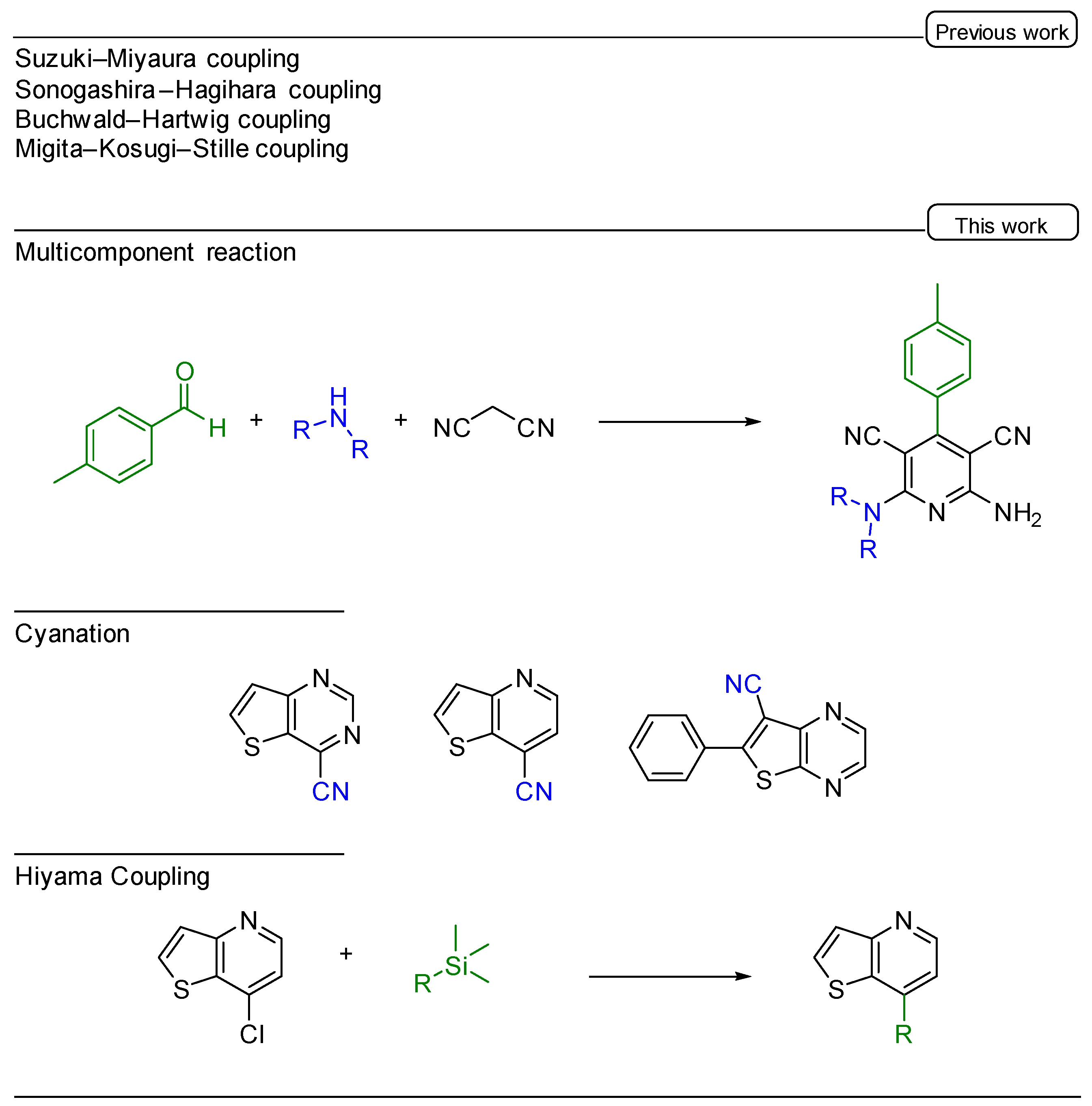
Catalysts | Free Full-Text | Eucalyptol: A Bio-Based Solvent for the Synthesis of O,S,N-Heterocycles. Application to Hiyama Coupling, Cyanation, and Multicomponent Reactions

Recyclable and Reusable Pd(OAc)2/XPhos–SO3Na/PEG-400/H2O System for Cyanation of Aryl Chlorides with Potassium Ferrocyanide | Catalysis Letters

Picolinaldehyde‐Zinc(II)‐Palladium(0) Catalytic System for the Asymmetric α‐Allylation of N‐Unprotected Amino Esters - Li - 2023 - Chemistry – A European Journal - Wiley Online Library
![Mechanisms of Catalyst Poisoning in Palladium-Catalyzed Cyanation of Haloarenes. Remarkably Facile C−N Bond Activation in the [(Ph3P)4Pd]/[Bu4N]+ CN- System | Journal of the American Chemical Society Mechanisms of Catalyst Poisoning in Palladium-Catalyzed Cyanation of Haloarenes. Remarkably Facile C−N Bond Activation in the [(Ph3P)4Pd]/[Bu4N]+ CN- System | Journal of the American Chemical Society](https://pubs.acs.org/cms/10.1021/ja078298h/asset/images/medium/ja078298hn00001.gif)
Mechanisms of Catalyst Poisoning in Palladium-Catalyzed Cyanation of Haloarenes. Remarkably Facile C−N Bond Activation in the [(Ph3P)4Pd]/[Bu4N]+ CN- System | Journal of the American Chemical Society

Nickel-Catalyzed Cyanation of Phenol Derivatives with Zn(CN)2 Involving C–O Bond Cleavage | The Journal of Organic Chemistry

Mild Palladium-Catalyzed Cyanation of (Hetero)aryl Halides and Triflates in Aqueous Media | Organic Letters

Recent developments and perspectives in palladium -catalyzed cyanation of aryl halides: synthesis of benzonitriles - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C1CS15004A

Recent developments and perspectives in palladium -catalyzed cyanation of aryl halides: synthesis of benzonitriles - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C1CS15004A
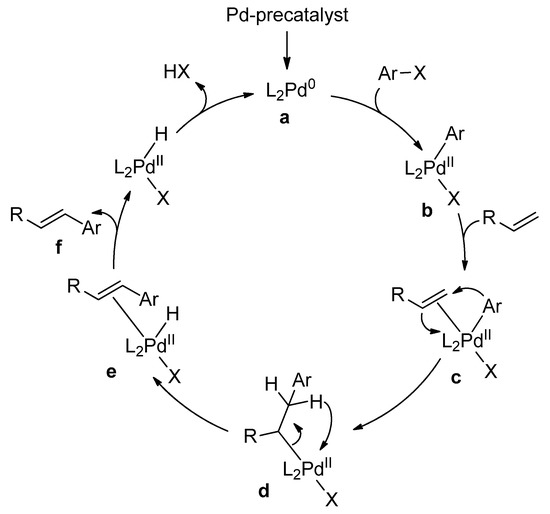
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond

Recent advances and prospects in the palladium-catalyzed cyanation of aryl halides - RSC Advances (RSC Publishing) DOI:10.1039/D0RA05960A

Table 2 from Improving Robustness: In Situ Generation of a Pd(0) Catalyst for the Cyanation of Aryl Bromides. | Semantic Scholar

Mild palladium-catalyzed cyanation of (hetero)aryl halides and triflates in aqueous media. - Abstract - Europe PMC







