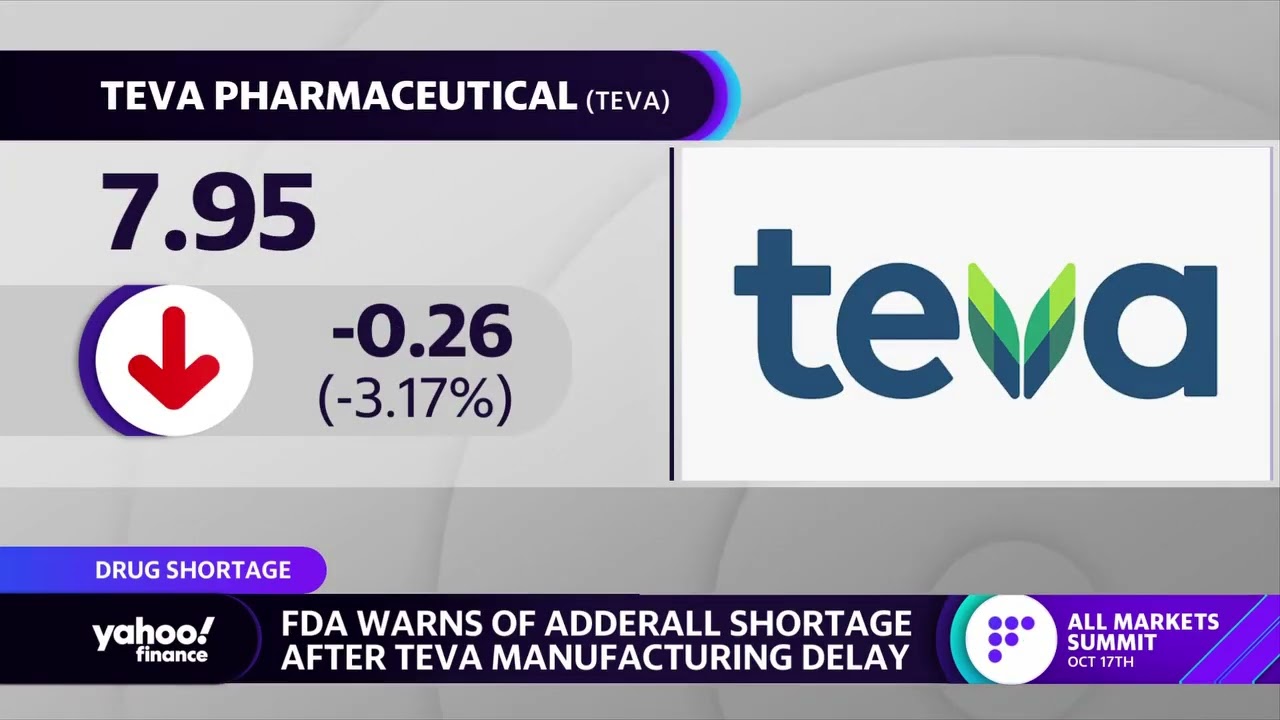
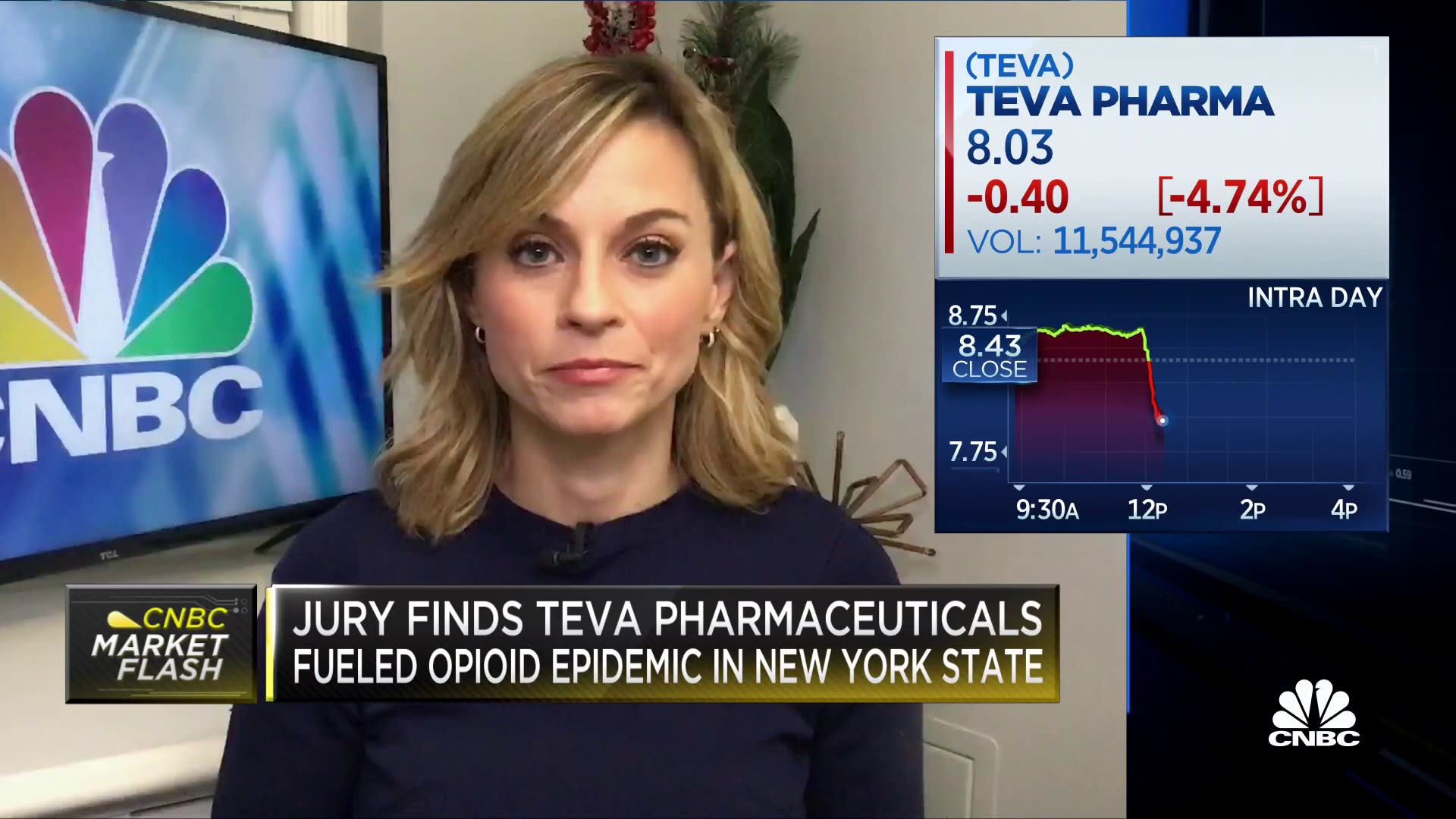
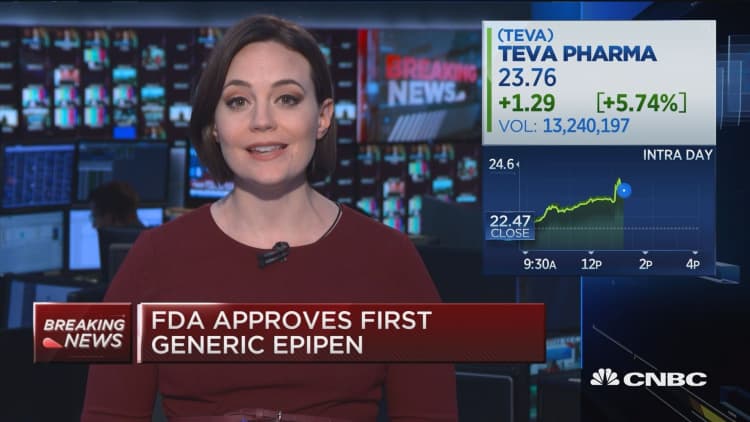
Teva Announces FDA Approval of First and Only Digital Inhaler with Built-In Sensors – ProAir® Digihaler™ | SnackSafely.com

Teva Pharmaceutical: Shares rise after FDA approves new migraine drug | Business News - The Indian Express

Teva Pharmaceuticals gets FDA approval for Osteoporosis injection | Eric A. Hughes, MD, PhD posted on the topic | LinkedIn

Teva Pharmaceuticals USA, Inc., Issues Voluntary Nationwide Recall of Specific Lots of Fentanyl Buccal Tablets CII Because of Labeling Error | ONS Voice

Teva Pharmaceuticals USA Issues Voluntary Nationwide Recall of all Amlodipine/Valsartan Combination Tablets and Amlodipine/Valsartan/Hydrochlorothiazide Combination Tablets that are Within Expiry | FDA