The five schemes of dividing the pyrrolidine ring structure using the... | Download Scientific Diagram

EP0365324A1 - Spiro-isoquinoline-pyrrolidine tetrones and analogs thereof useful as aldose reductase inhibitors - Google Patents

Azetidiniums: Ring‐Expansion to Pyrrolidines, Piperidines, Azepanes, and Azocanes - Masson - 2020 - European Journal of Organic Chemistry - Wiley Online Library
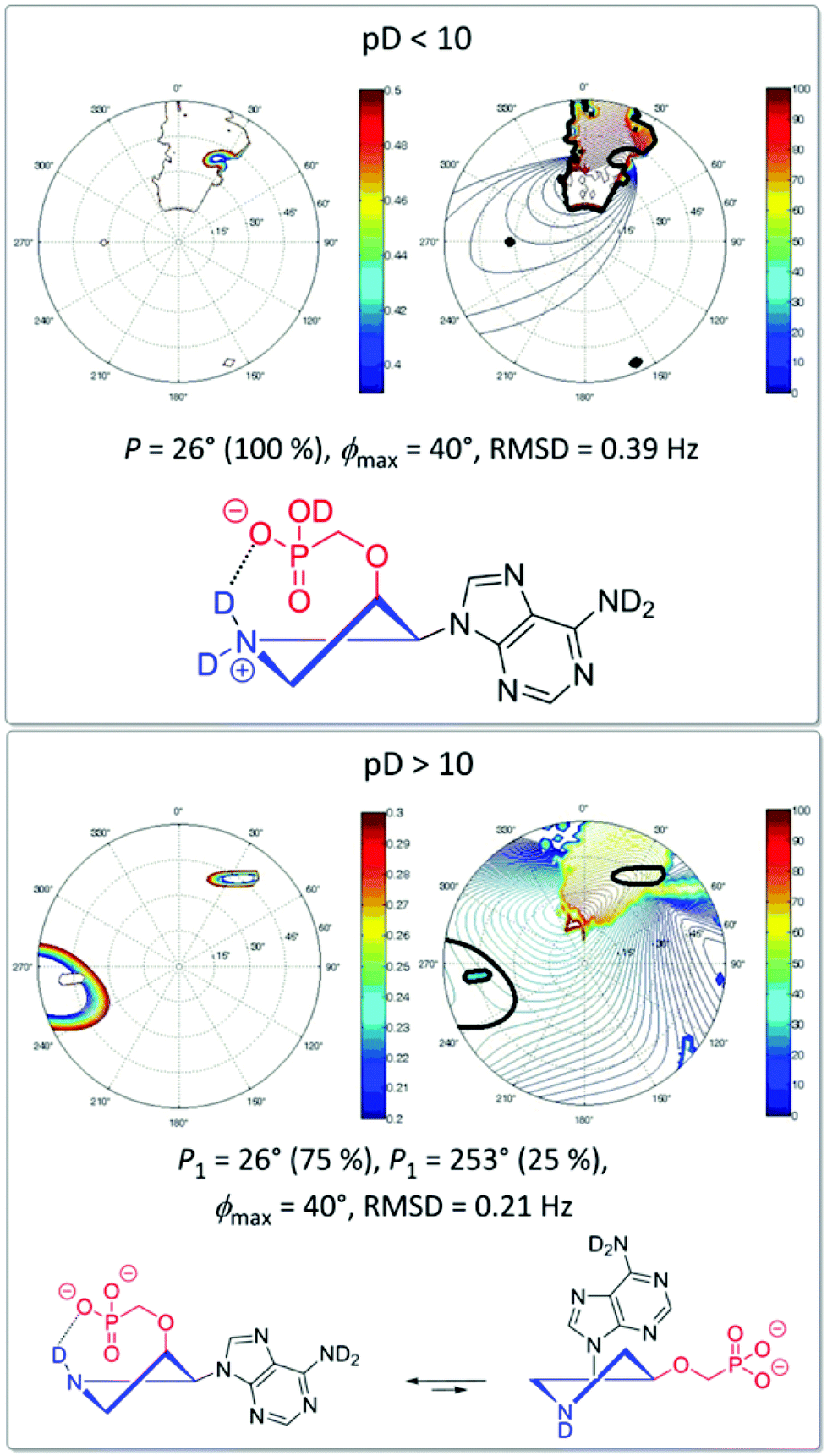
Synthesis, conformational studies, and biological properties of phosphonomethoxyethyl derivatives of nucleobases with a locked conformation via a pyrr ... - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB00097A

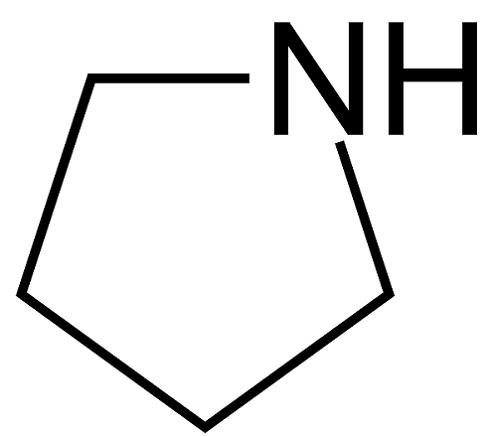
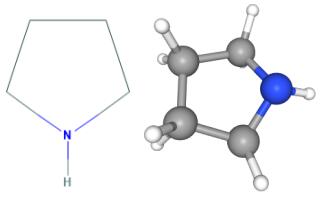
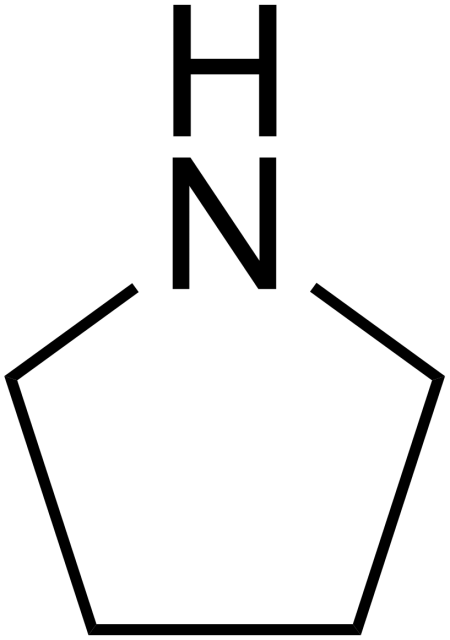
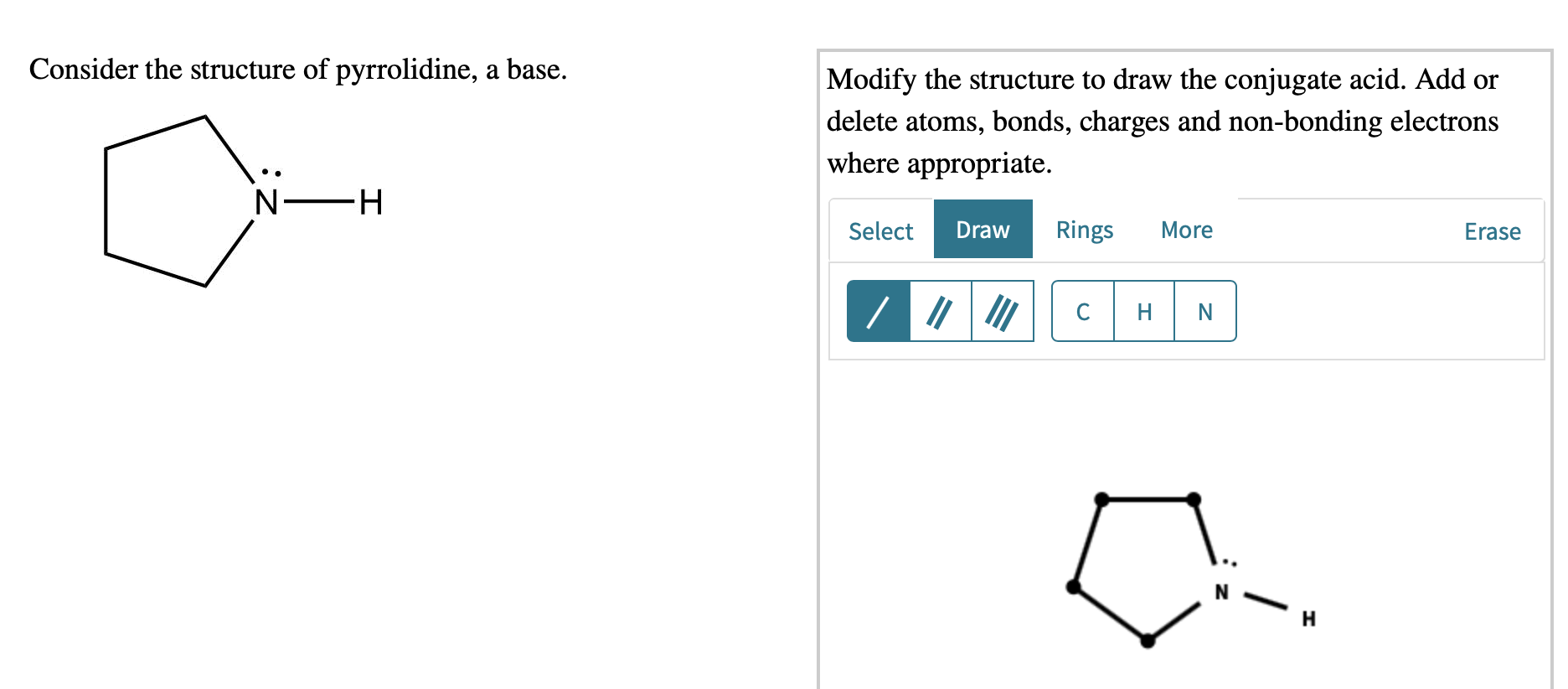
Químicos Orgánicos de México - Pyrrolidine is the cyclic amine with the formula (CH2)4NH. This colorless liquid has an odor described as shellfish-like or semen-like. Pyrrolidine is produced industrially by catalytic cyclization
![Spiro[pyrrolidine-3, 3´-oxindole] as potent anti-breast cancer compounds: Their design, synthesis, biological evaluation and cellular target identification | Scientific Reports Spiro[pyrrolidine-3, 3´-oxindole] as potent anti-breast cancer compounds: Their design, synthesis, biological evaluation and cellular target identification | Scientific Reports](https://media.springernature.com/m685/springer-static/image/art%3A10.1038%2Fsrep32213/MediaObjects/41598_2016_Article_BFsrep32213_Fig1_HTML.jpg)
Spiro[pyrrolidine-3, 3´-oxindole] as potent anti-breast cancer compounds: Their design, synthesis, biological evaluation and cellular target identification | Scientific Reports

A Flavin-Dependent Oxygenase Catalyzes Hydroxylation and Simultaneous Pyrrolidine Ring Formation in Protubonine Biosynthesis in Aspergillus ustus | Journal of Natural Products

Numbering of pyrrolidine's ring in N-acetylproline. The bond-cutting... | Download Scientific Diagram

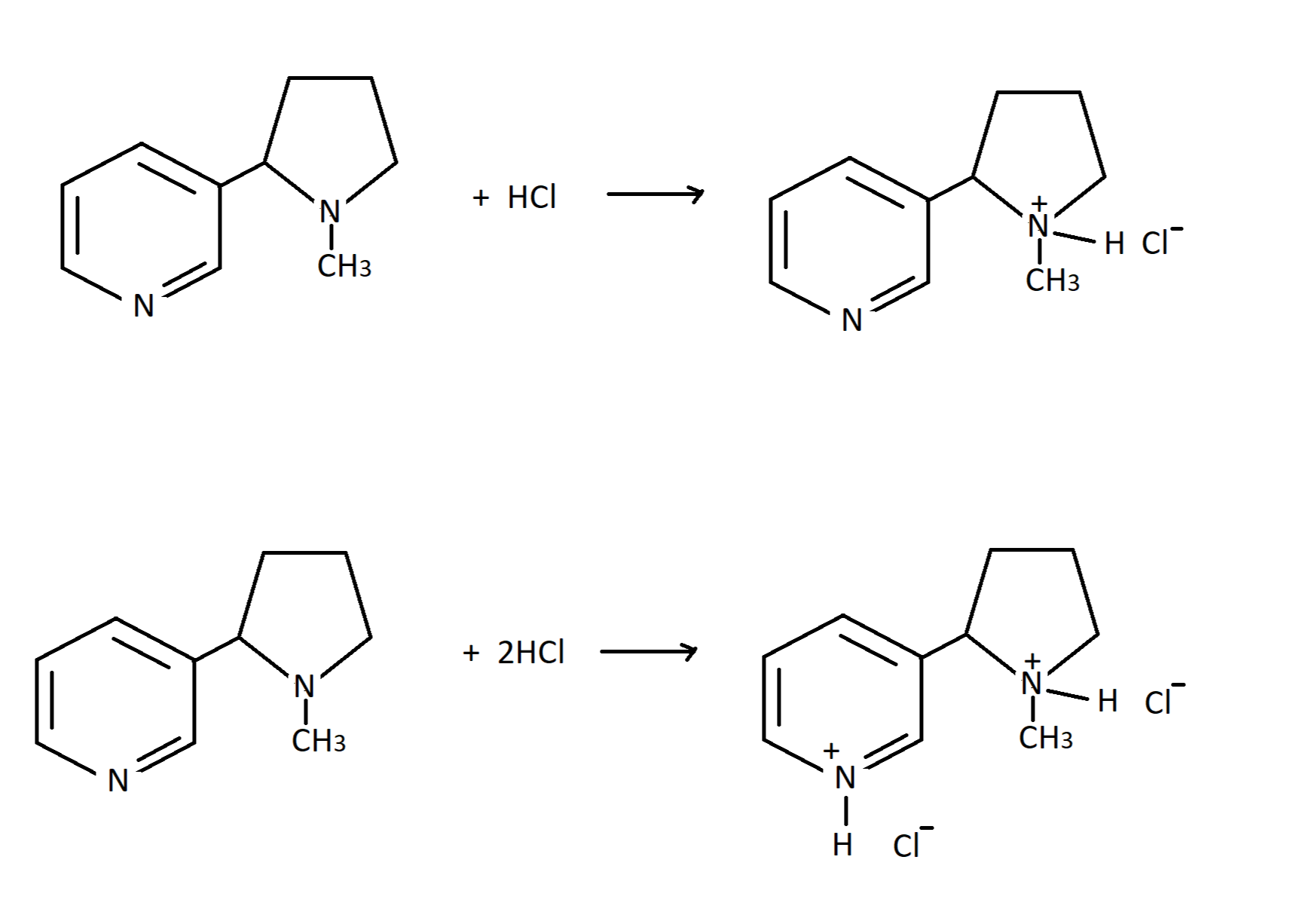
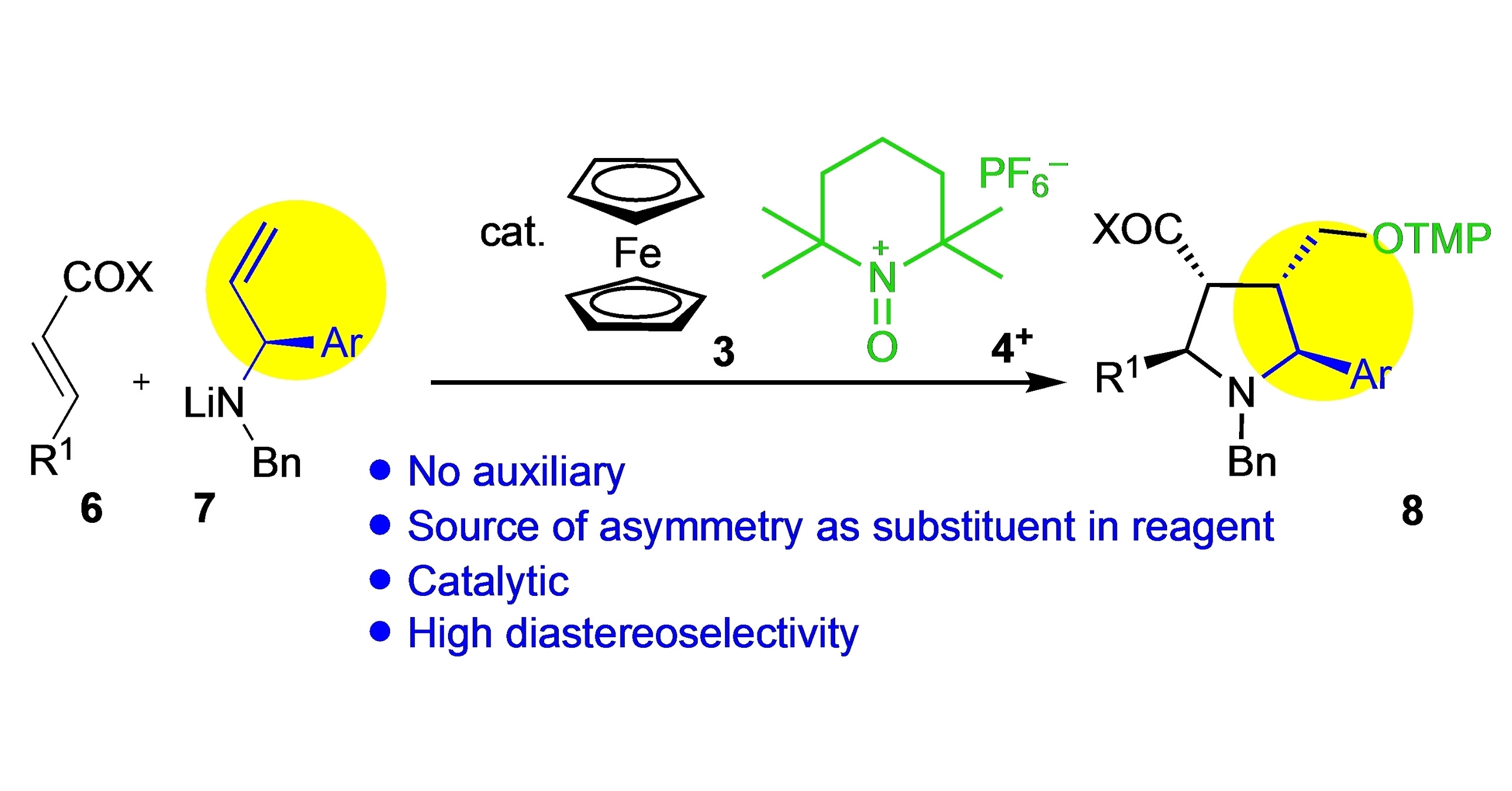
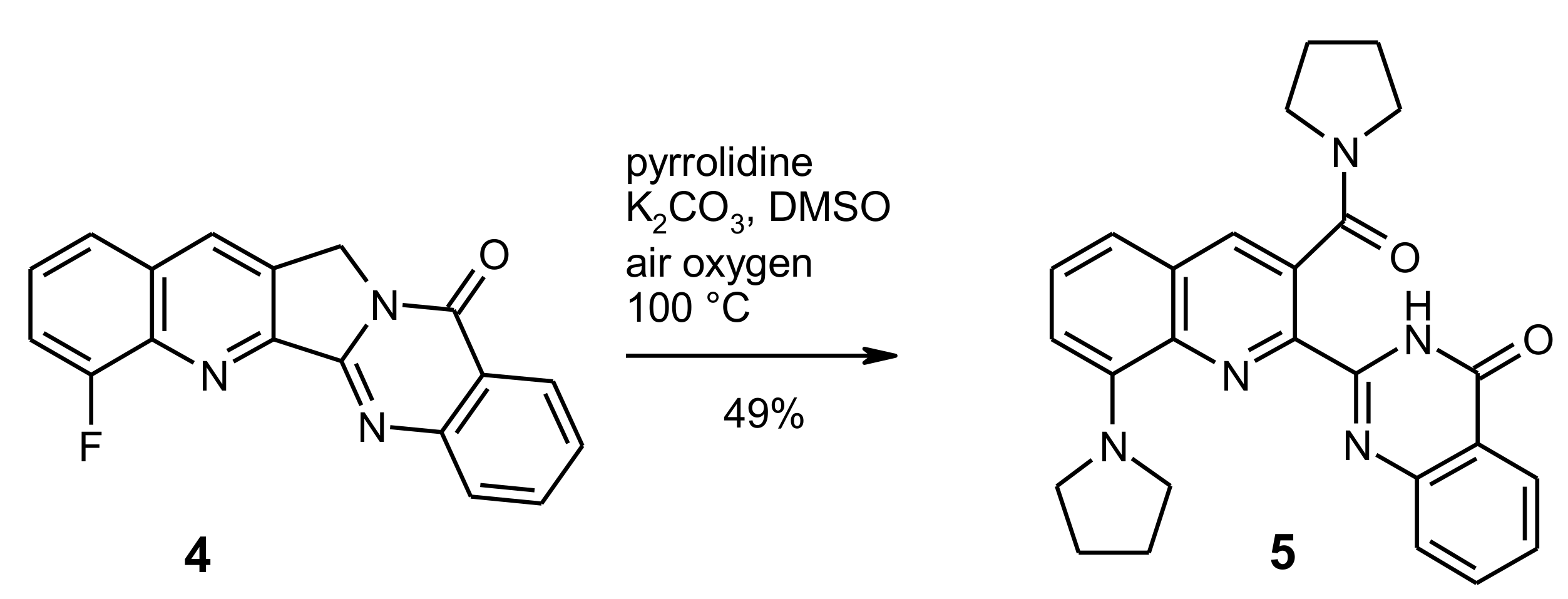
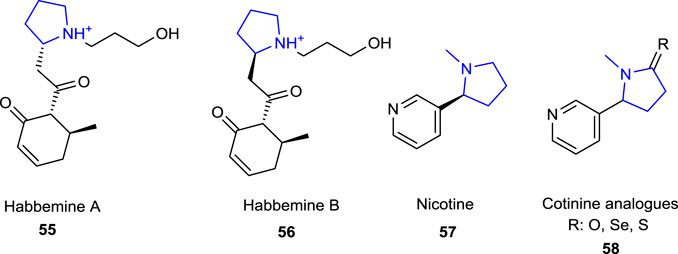
Pyrrolidine in Drug Discovery: A Versatile Scaffold for Novel Biologically Active Compounds | Topics in Current Chemistry