Palladium-Catalyzed Arylation of Malonates and Cyanoesters Using Sterically Hindered Trialkyl- and Ferrocenyldialkylphosphine Ligands | The Journal of Organic Chemistry

Palladium-Catalyzed Arylation of Malonates and Cyanoesters Using Sterically Hindered Trialkyl- and Ferrocenyldialkylphosphine Ligands | The Journal of Organic Chemistry

Pd(0)-Catalyzed Asymmetric 7-Endo Hydroacyloxylative Cyclization of 1,6-Enyne Enabled by an Anion Ligand-Directed Strategy | Journal of the American Chemical Society
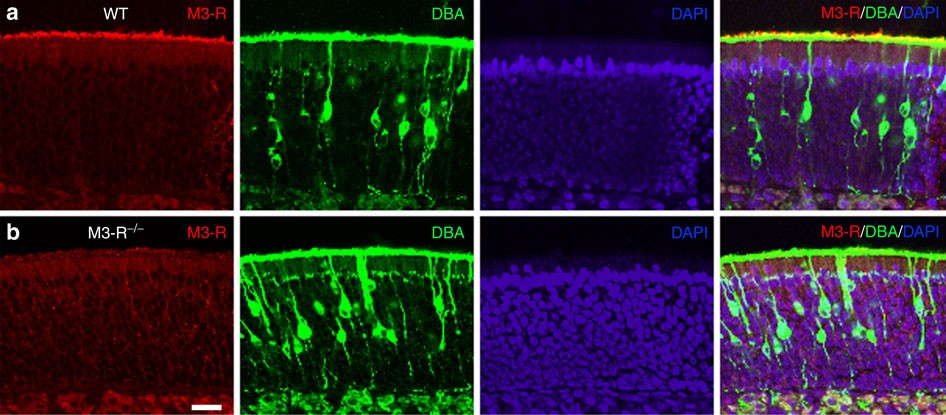
Muscarinic acetylcholine receptor M3 modulates odorant receptor activity via inhibition of β-arrestin-2 recruitment | Nature Communications