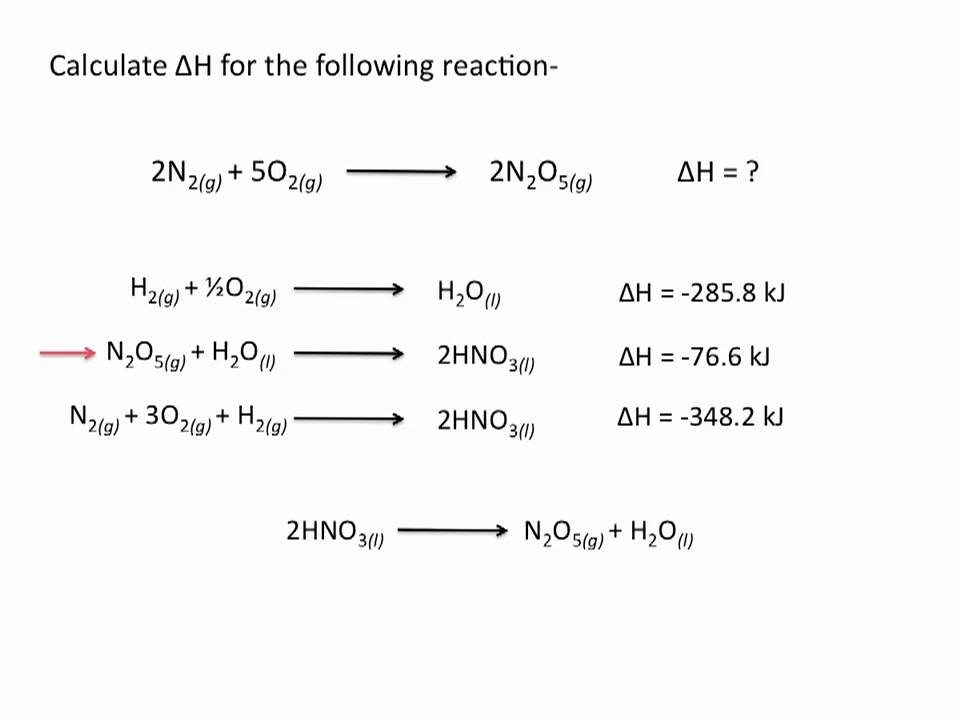
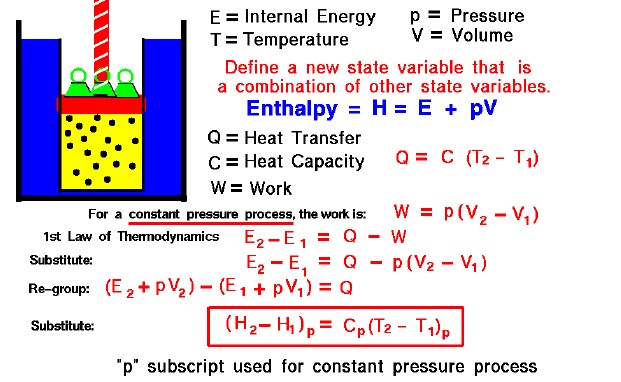
thermodynamics - Calculating Enthalpy of formation versus Calculating Enthalpy of a reaction not occurring at standard conditions - Chemistry Stack Exchange

Hess's law A -> B Delta H= +30kJ B -> C Delta H= +60kJ A.) Use Hess's law to calculate the - brainly.com




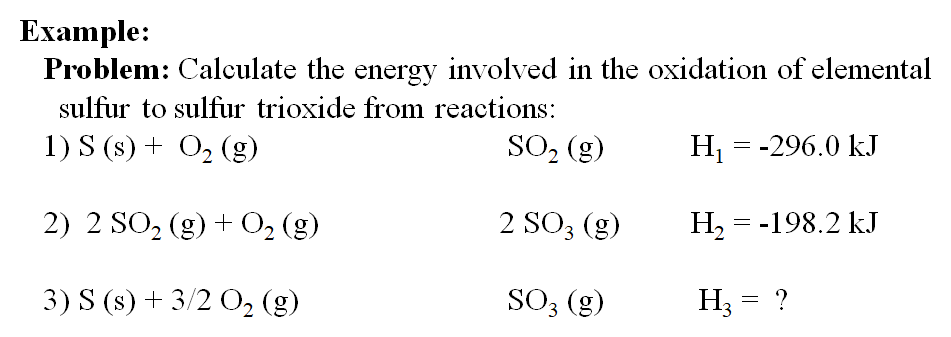


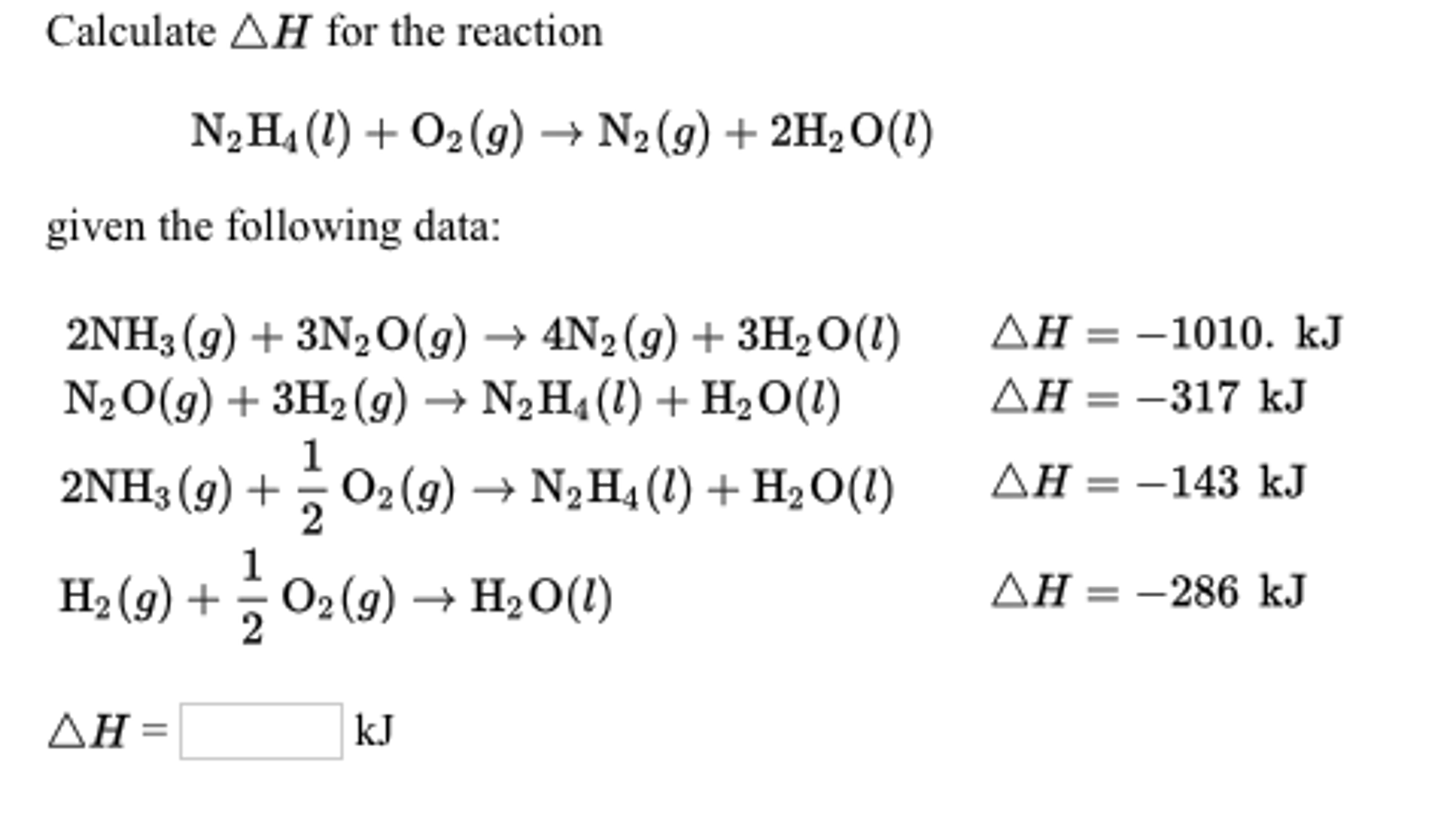




![5.1 Delta Hf and Delta Hc calculations [SL IB Chemistry] - YouTube 5.1 Delta Hf and Delta Hc calculations [SL IB Chemistry] - YouTube](https://i.ytimg.com/vi/zMksewz0cNU/maxresdefault.jpg)