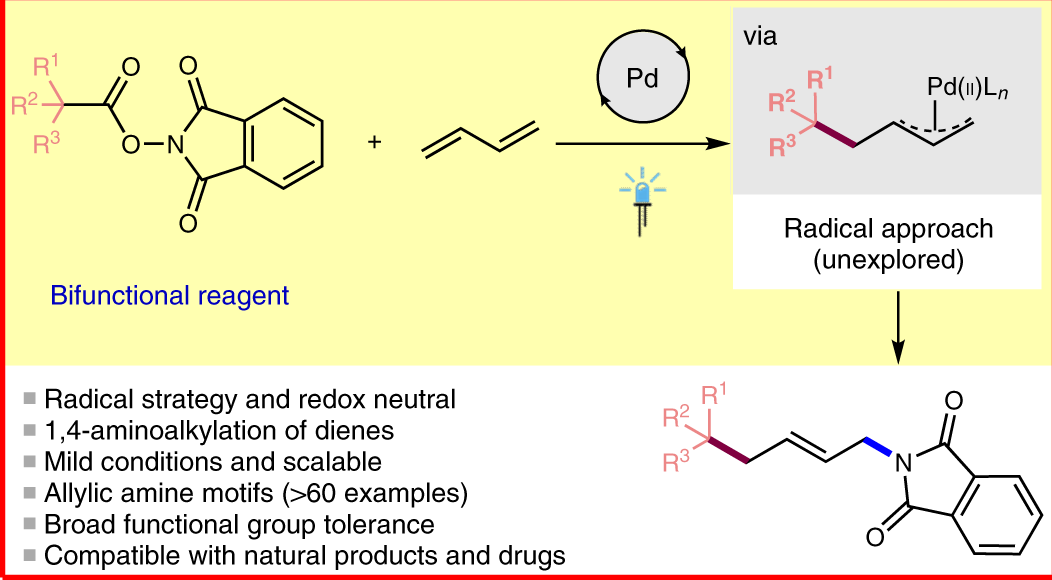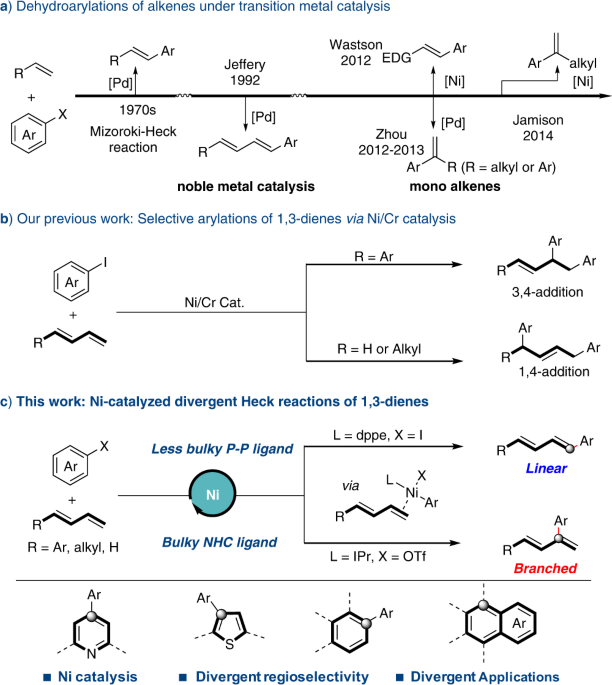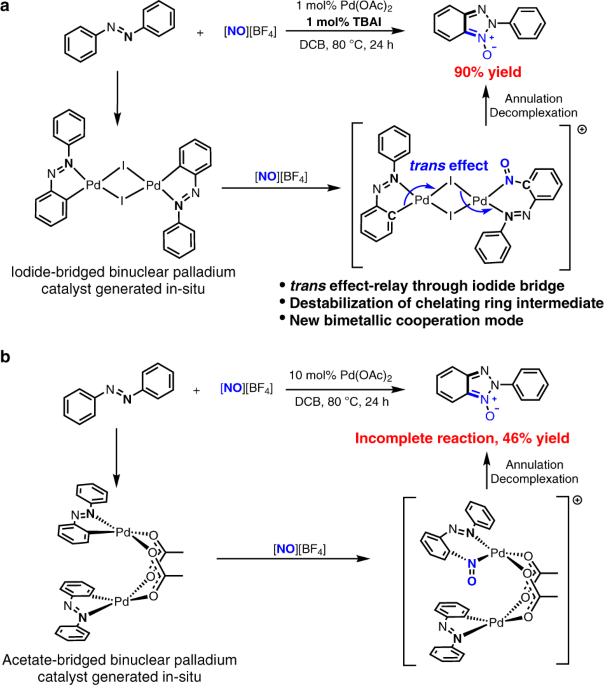
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry
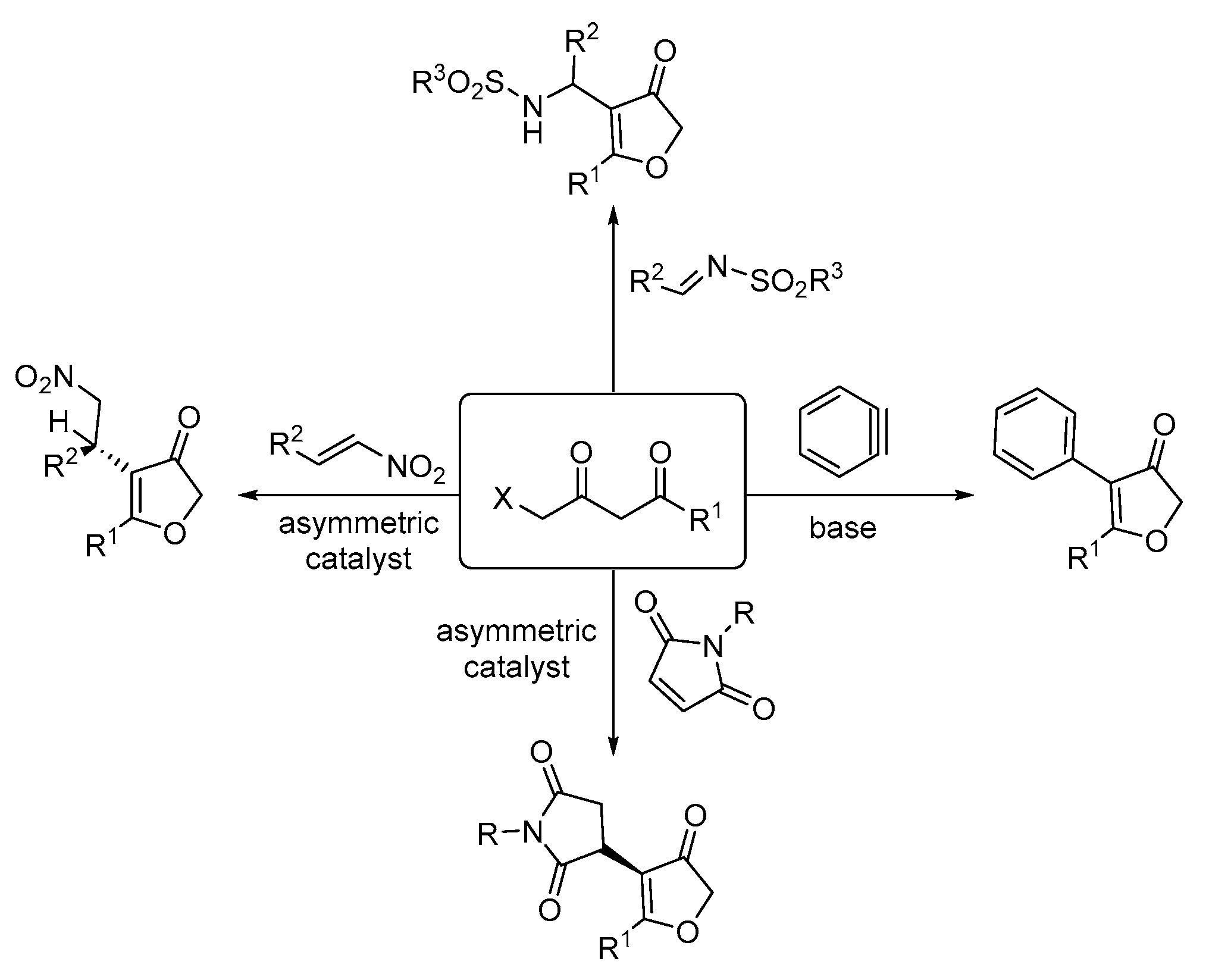
Organics | Free Full-Text | Palladium Catalyzed Ring-Opening of Diazabicylic Olefins with 4-Halo-1,3-Dicarbonyl Compounds: Accessing 3(2H)-Furanone-Appended Cyclopentenes
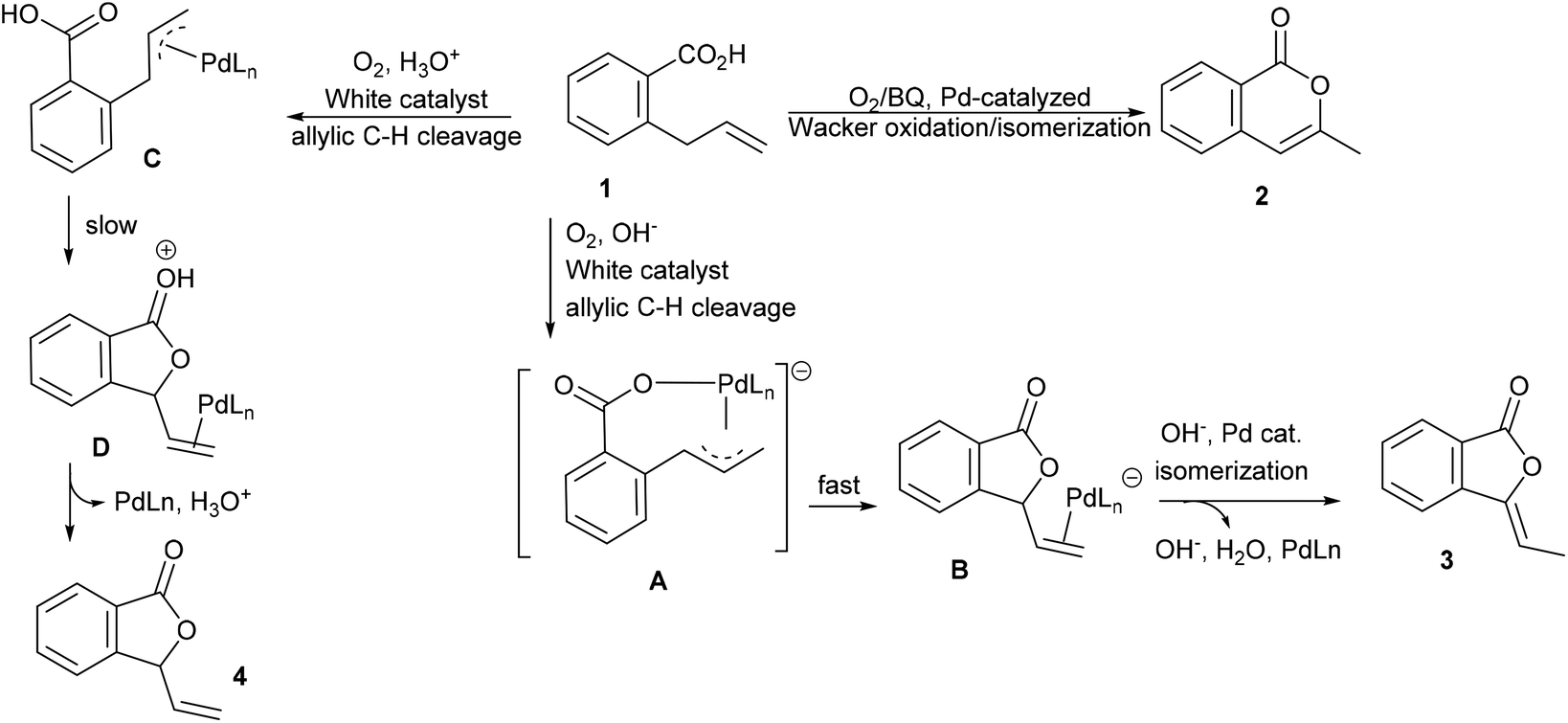
Acid–base-sensitive allylic oxidation of 2-allylbenzoic acids to form phthalides - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D0OB00303D

Palladium‐Catalyzed Allyl Cross‐Coupling Reactions with In Situ Generated Organoindium Reagents - Lee - 2011 - Chemistry – An Asian Journal - Wiley Online Library

Recent Expedition in Pd‐Catalyzed Sonogashira Coupling and Related Processes† - Choy - 2023 - Chinese Journal of Chemistry - Wiley Online Library

The role of allyl ammonium salts in palladium-catalyzed cascade reactions towards the synthesis of spiro-fused heterocycles | Nature Communications

Palladium-Catalyzed γ-Selective and Stereospecific Allyl−Aryl Coupling between Acyclic Allylic Esters and Arylboronic Acids | Journal of the American Chemical Society

Scope and Mechanism of Allylic C−H Amination of Terminal Alkenes by the Palladium/PhI(OPiv)2 Catalyst System: Insights into the Effect of Naphthoquinone | Journal of the American Chemical Society

Scheme 3. Part of the mechanism for the palladium catalyzed allylic... | Download Scientific Diagram

Palladium-catalyzed cross-coupling of five-membered heterocyclic silanolates. - Abstract - Europe PMC

Palladium-Catalyzed γ-Selective and Stereospecific Allyl−Aryl Coupling between Acyclic Allylic Esters and Arylboronic Acids | Journal of the American Chemical Society

Palladium-Catalyzed Reductive Aminocarbonylation of o-Iodophenol-Derived Allyl Ethers with o-Nitrobenzaldehydes to 3-Alkenylquinolin-2(1H)-ones | Organic Letters

Palladium‐Catalyzed Allyl Cross‐Coupling Reactions with In Situ Generated Organoindium Reagents - Lee - 2011 - Chemistry – An Asian Journal - Wiley Online Library

Palladium-catalyzed direct CH allylation of indoles derivatives with allyltrimethylsilanes - ScienceDirect

Palladium-catalysed branch- and enantioselective allylic C–H alkylation of α-alkenes | Nature Synthesis

Palladium-Catalyzed γ-Selective and Stereospecific Allyl−Aryl Coupling between Acyclic Allylic Esters and Arylboronic Acids | Journal of the American Chemical Society